|
Once we get all the values of entropy change, just add them up to get the standard molar entropy of vaporization of water. This was the best explanation for this problem! I just followed these steps and finally got the right answer :) The standard state of a substance is an arbitrarily. The molar integral entropy of adsorption Sa, can be defined as Sa Sa Sg (2.8) where Sa is the mean molar entropy of all the adsorbed molecules over. In the statement 'all spontaneous changes result in an increase in entropy', the entropy change is. Standard molar entropy is the amount of entropy one mole of substance has under its standard state. Just plug and solve to get that entropy change Which of the following has the largest standard molar entropy B) H (g) Which of the following best describes the reaction 2 HS (g) + 3 O (g) 2 HO (g) + 2 SO (g) B) S has a small negative value. Standard molar entropy, S o liquid: 200.4 J/(mol K) Enthalpy of combustion, c H o 1785.7 kJ/mol Heat capacity, c p: 125.5 J/(mol K) Gas properties Std enthalpy change of formation, f H o gas: 218.5 kJ/mol Standard molar entropy, S o gas: 295.35 J/(mol K) Heat capacity, c p: 75 J/(mol K) van der Waals' constants: a 1409.4 L 2 kPa. Only this time, we use the Cpm 33.6 J⋅K−1⋅mol−1 for water vapor and the temperature change is from 100 ☌ to 59 ☌. To solve the third part, we use again the equation ∆S = Cpm ln (T2/T1). Since the Cpm is 75.3 J⋅K−1⋅mol−1 for liquid water and the temperature change is from 59.0 ☌ to 100 ☌, just plug and solve to get the entropy changeĪs for the second part, we already are given the entropy of vaporization of water at 100 ☌ which is 109.0 J⋅K−1⋅mol−1. To solve the first part, we should use the equation ∆S = Cpm ln (T2/T1). Entropy 2 Assignment SCH4U - Grade 12 University prep Chemistry (Ontario. For any chemical reaction, the standard entropy change is the sum of the standard molar entropies of the products minus the sum of the standard molar entropies of the reactants. Calculate the molar standard entropy S(T,p) (J/molK) under H water 2(g) vapour the at. Worksheet- Balancing Redox Reactions Using Half Rxns. The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. 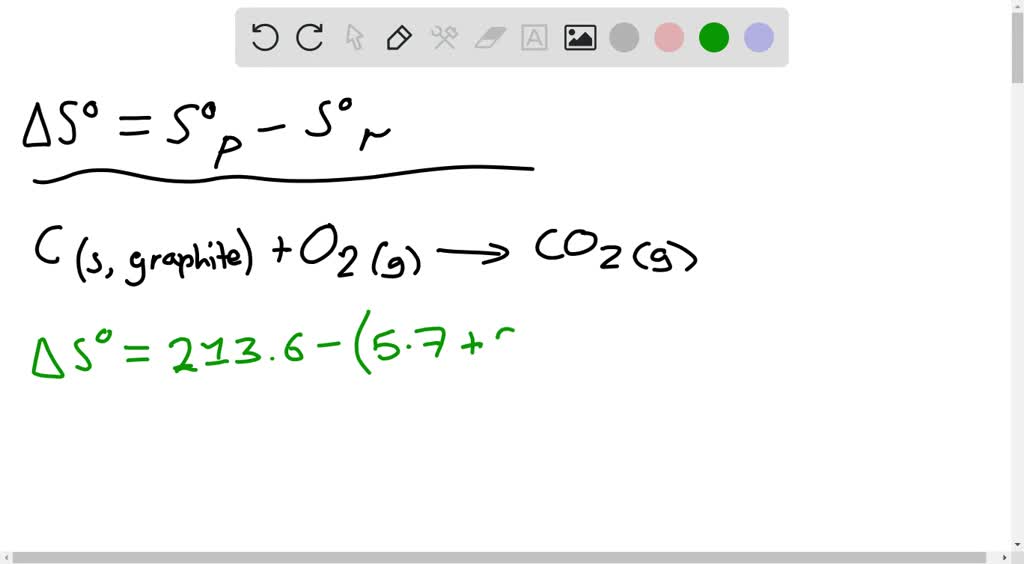
A table like this can be used in much the same way as a table of standard enthalpies of formation in order to find the entropy change Sm for a reaction occurring at standard pressure and at 298 K. To solve this problem, we need to know three steps that involve finding the standard molar entropy of vaporization: the entropy change heating water from 59.0 ☌ to 100 ☌, the entropy of vaporization of water at 100.0 ☌, and the entropy change cooling water from 100 ☌ to 59 ☌. Calculate the change of entropy S (J/K) in the total system when. Values of the standard molar entropies of various substances at 298 K (25C) are given in the table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
